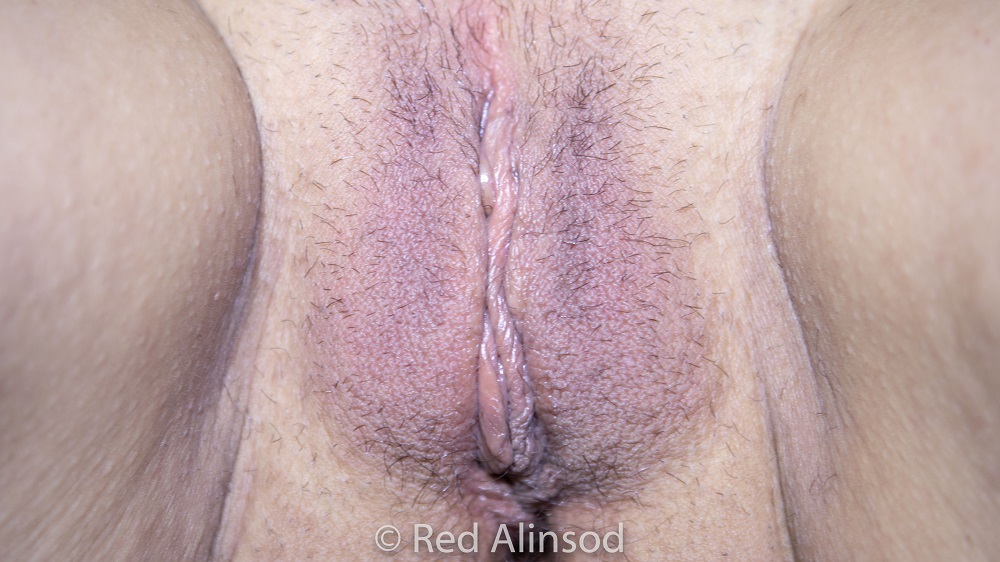
Laxity of Skin
Case 16
PATIENT HISTORY:
thermiva case studyPatient in her early 50s complains of vulvar laxity, vaginal dryness, repetitive urinary and vaginal infections, and a leaky bladder. She underwent ThermiVa treatments and was placed on vaginal estrogen therapy which resolved her symptoms completely.
Results: Her stress incontinence and overactive bladder and nocturia were eliminated.