Loose Skin
(Section 3)
Case 11
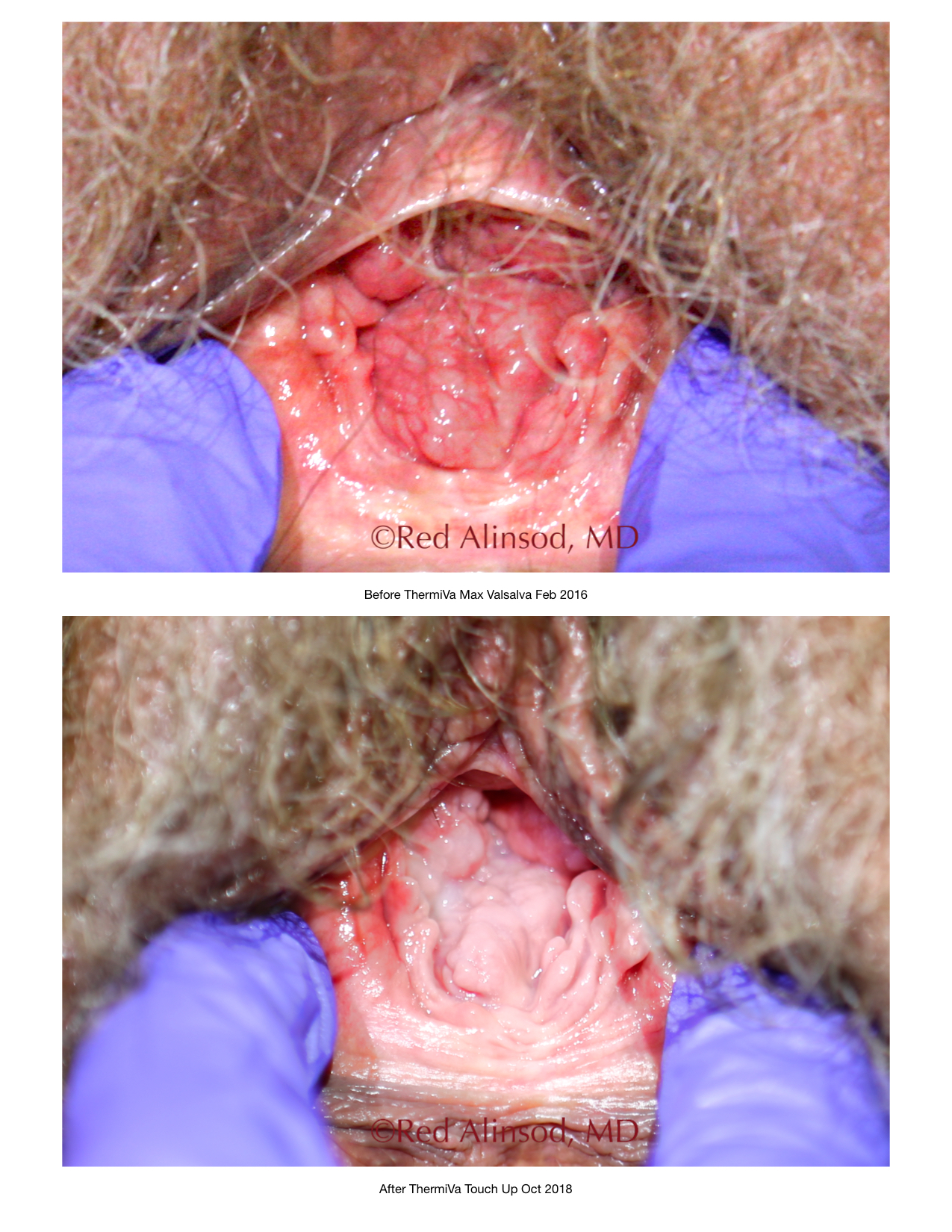 ThermiVa Over 2 Years
ThermiVa Over 2 Years
 ThermiVa Over 2 Years
ThermiVa Over 2 Years
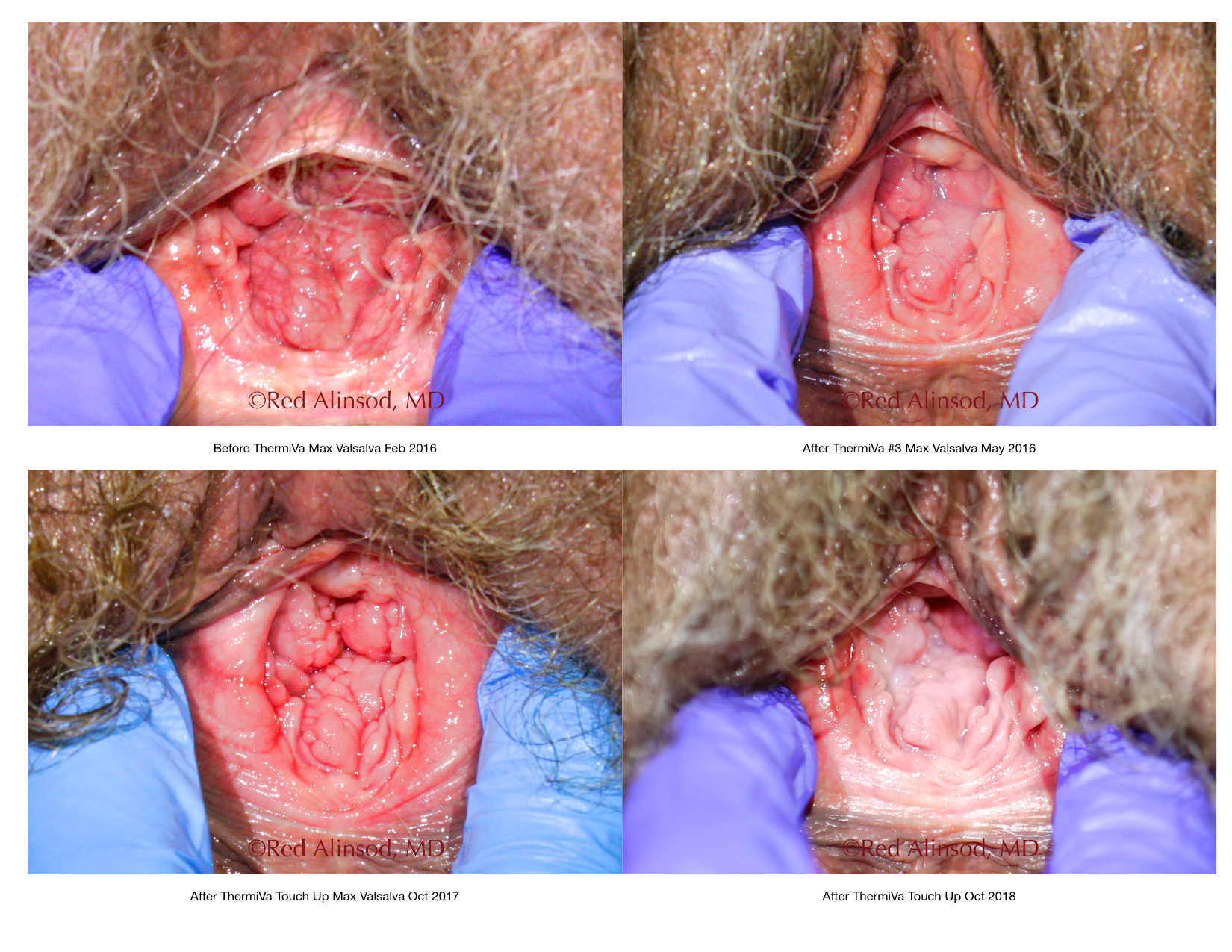
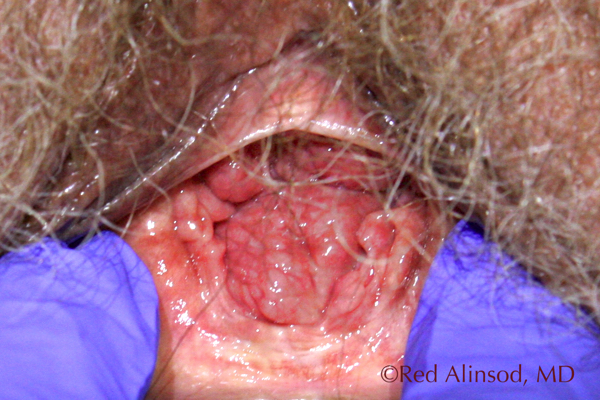 Before ThermiVa Max Valsalva
Before ThermiVa Max Valsalva
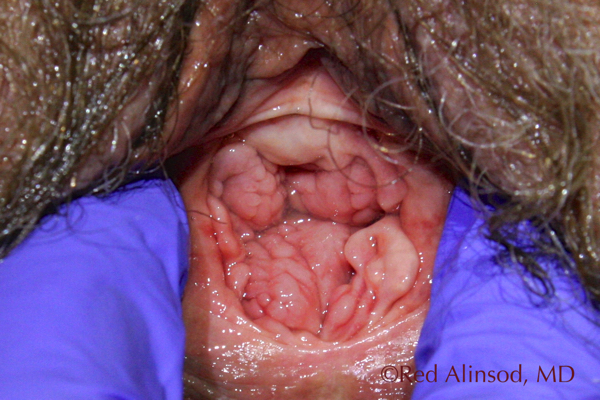 After ThermiVa Max Valsalva
After ThermiVa Max Valsalva
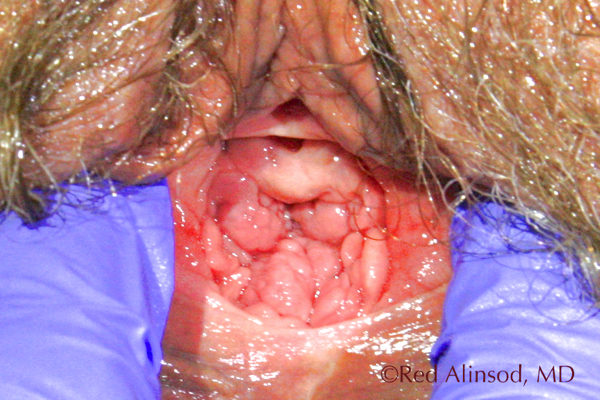 After ThermiVa Max Valsalva
After ThermiVa Max Valsalva
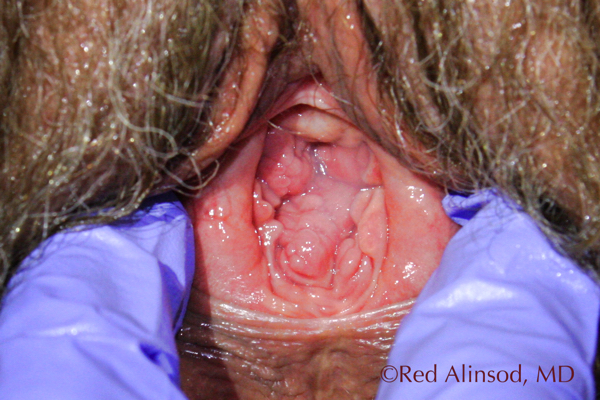 After ThermiVa Max Valsalva
After ThermiVa Max Valsalva
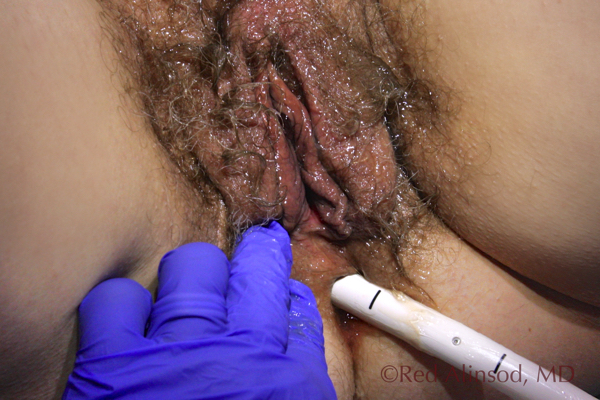 Transanal Treatment
Transanal Treatment
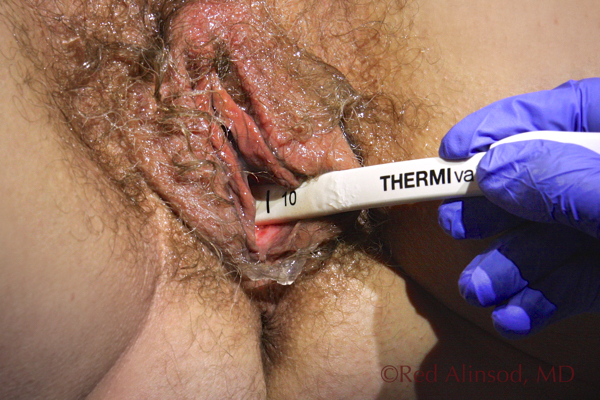 Transvaginal Treatment
Transvaginal Treatment
 ThermiVa Over 2 Years
ThermiVa Over 2 Years
 ThermiVa Over 2 Years
ThermiVa Over 2 Years
 Before ThermiVa Max Valsalva
Before ThermiVa Max Valsalva
 After ThermiVa Max Valsalva
After ThermiVa Max Valsalva
 After ThermiVa Max Valsalva
After ThermiVa Max Valsalva
 After ThermiVa Max Valsalva
After ThermiVa Max Valsalva
 Transanal Treatment
Transanal Treatment
 Transvaginal Treatment
Transvaginal Treatment